Why Partner With Us?
Specialized Research Facilities
Specialized Research Facilities
Specialized Research Facilities
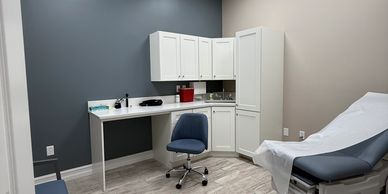
OCR features a dedicated Sleep Lab, Echo, PFT capabilities, and a high-volume Lab Draw Station within a modern, fully equipped environment. Our facility is designed to support Phase II-IV trials with the precision required for high-quality data collection.
Multidisciplinary Clinical Team
Specialized Research Facilities
Specialized Research Facilities

Our staff consists of board-certified Principal Investigators and coordinators with extensive experience in family medicine and complex trial oversight. We provide a patient-centric environment that ensures high retention and strict protocol adherence.
Sponsor-First Operations
Specialized Research Facilities
Therapeutic Depth & Diversity

We prioritize the "business of science" by offering CROs streamlined feasibility, rapid regulatory startup, and transparent communication. Our site utilizes advanced EMR systems to provide real-time updates and seamless remote monitoring.
Therapeutic Depth & Diversity
Therapeutic Depth & Diversity

We deliver immediate access to diverse, underrepresented populations—including African American, Hispanic, and rural geriatric communities—vital for meeting FDA mandates. Integrated with a high-volume family practice, we specialize in COPD, Heart Failure, Diabetes, and Obesity, alongside high-demand indications like NASH, Osteoarthritis, CKD, and Vaccines.
Accelerated Patient Enrollment
Regulatory & Quality Excellence
By leveraging a primary care practice with 15,500+ active patients and 215+ daily visits, we eliminate referral lag. This internal database allows us to identify treatment-naive subjects and hit enrollment targets weeks ahead of schedule.
Regulatory & Quality Excellence
Regulatory & Quality Excellence
Accuracy is the cornerstone of our operations; our staff is 100% CITI/GCP certified and proficient in EDC and IRT systems. We strictly adhere to ALCOA+ data standards, ensuring every data point is audit-ready for the FDA.
This website uses cookies.
We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.